Integrated Biopharma Engineering & Equipment — Built First Time Right
ZETA combines process engineering, automation, and precision fabrication under one roof. Our integrated approach reduces risk, shortens time-to-market, and ensures your system performs exactly as intended — the first time.
Process Systems for Today’s Biopharma Challenges

Modern biopharma manufacturing is complex – from mAbs to vaccines to plasma products, every modality brings unique risks and timetables. ZETA helps you bridge the gap between process design and GMP production, with systems engineered and fabricated so your equipment is GMP-ready on day one – without rework or surprises.
Applications we support include:
- Monoclonal antibodies (mAbs) & ADCs - shorten tech transfer with integrated upstream and downstream workflows and high‑containment systems that tackle the complexity of mAbs and ADC manufacturing.
- Vaccines, parenterals & plasma products - accelerate delivery with precision systems supporting all stages of blood plasma fractionation and aseptic processing with cold‑chain solutions for vaccine and parenteral manufacturing.
- Cell & gene therapies and mRNA - manage scale‑up and scale‑out with GMP‑compliant infrastructure, digital tools, and scalable, controlled production solutions that enhance therapeutic development.
- Recombinant proteins & peptides - streamline production and reduce costs with scalable, optimized upstream and downstream processes for recombinant proteins and peptides.
200+
Biopharma Plants Worldwide
1200+
Engineers on Staff
35+
Years in Business
500+
Skids in Use
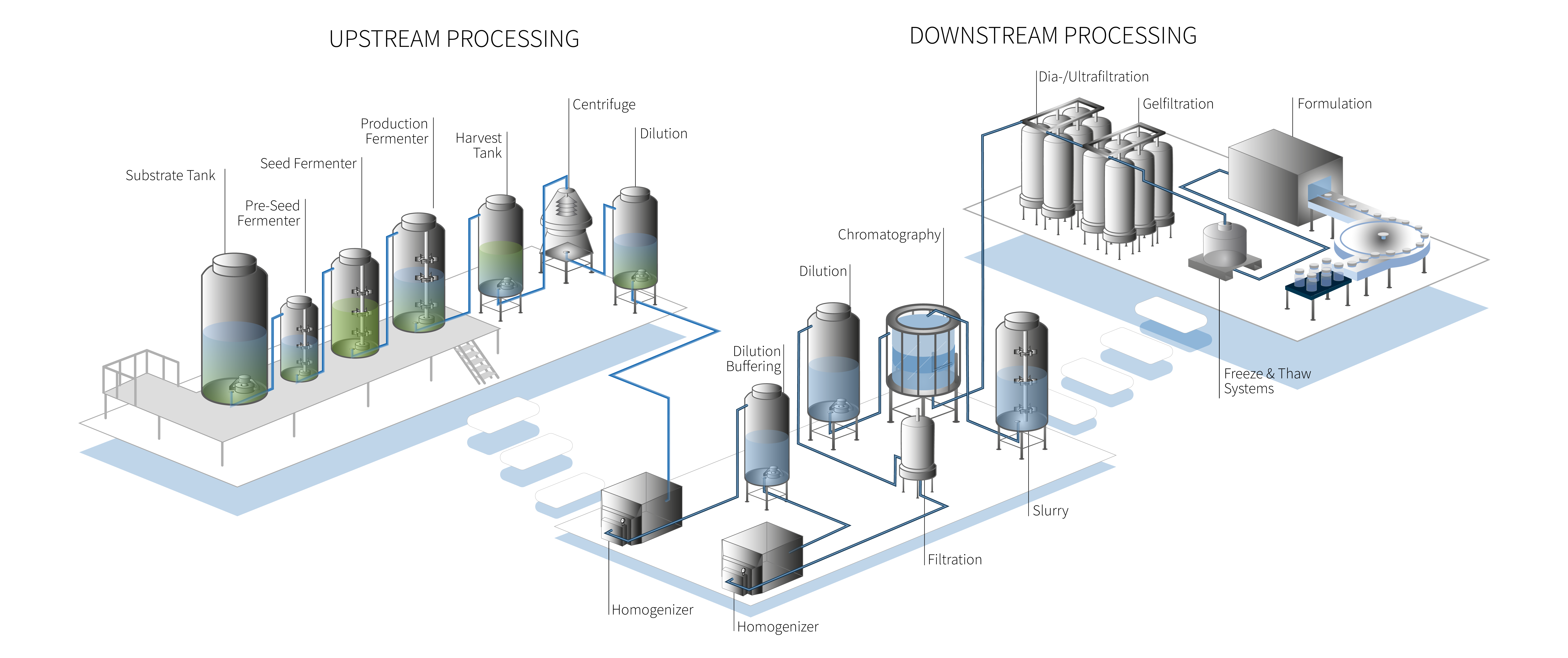
Biopharma Value Chain: From Upstream to Final Formulation
ZETA designs and fabricates process systems across upstream, downstream, preparation, and formulation — delivering integrated, automated solutions that fit seamlessly into your facility.
Confident Projects. Faster to Market
Modern biotech facilities can’t afford costly redesigns or delays. Whether you’re a biotech manufacturer scaling production or an A&E partner designing the facility, success depends on systems that perform — without rework or surprises.
With ZETA, you gain an integrated partner that unites engineering, automation, and fabrication under one roof. From early process simulation through delivery, our systems are designed to accelerate your path to GMP production.
- Reduce risk with process simulation before CapEx is committed.
- Integrated design + fabrication means fewer handovers and fewer surprises.
- Turnkey skids and super-skids delivered fully GMP-compliant — no rework, no delays.
- Precision fabrication and full digital traceability ensure your equipment is audit-ready from day one.
ZETA’s team supports both biotech manufacturers and their A&E partners with specialized process expertise, simulation-backed designs, and a 10-step fabrication process that ensures quality at every stage — from incoming material checks to dynamic FAT.
Our collaborative approach helps A&E partners strengthen bids and deliver higher-value facilities, while giving end customers confidence in every skid delivered.
Subscribe for Insights
Get the latest articles, case studies and event updates from ZETA's biotech engineering specialists - covering process equipment, modular skids, and integrated system design for smarter, faster, first-time-right project delivery.
ZETA Newsletter signup
Real-World Examples & Expert Insights
ZETA combines digital simulation, precision fabrication, and GMP expertise to help biotech manufacturers and their A&E partners deliver safe, pure, and effective production systems. Explore how the approach translates from design to delivery:

Case Study: Super-Skid Bioreactors for mAb Production
Integrated engineering and fabrication cut project timelines and eliminated rework for a large-scale mAb facility — ZETA’s “first-time-right” approach in action.

White Paper: Predictive Bioreactor Characterization for Reliable Scale-Up
Explore how digital twins and process simulation predict bioreactor behavior with precision, reducing uncertainty long before CapEx is committed.

Case Study: Buffer Inline Conditioning Skid for Process Efficiency
A closer look at InFlow, ZETA’s just-in-time buffer system that minimizes facility footprint, streamlines changeovers, and boosts downstream flexibility.
Fabrication, Reinvented with Digital Integration
Most suppliers focus only on the steps of fabrication. ZETA’s approach is different: every stage is digitally integrated and continuously verified, so quality is built in from the start.
- Integrated design + fabrication - Engineering changes flow automatically across disciplines, preventing mismatches between design and build.
- Automated compliance checks - Materials, surface finishes, and certifications are validated before fabrication begins
- Full digital traceability - Every weld, component, and document is tracked in real time — reducing manual entry, eliminating rework, and simplifying audits.
The result is more than equipment — it’s a digitally assured, GMP-compliant process that gives you confidence in every skid delivered.
Learn with ZETA — Schedule a Lunch & Learn
ZETA’s experts share practical insights on biopharma facility design and process equipment. These Lunch & Learn sessions are educational and no-obligation—they’re tailored for your team and delivered in a convenient format, virtual or on‑site. We focus on exchanging knowledge, not pitching products, so you can tap into decades of experience and leave with actionable ideas.
Featured Topics:
Typical Timelines for Our Biopharma Process Systems
-
Biopharma projects require careful planning and realistic timelines. While every system is tailored to your process, the ranges below provide a general view of how ZETA delivers complex process systems at scale.
Typical Lead Time (PO to Delivery)
| Filtration & purification systems (UF/DF, TFF) | 52 weeks |
| Bioreactor & process vessel skids (including media & buffer prep) | 56 weeks |
| Inline conditioning systems (on-demand buffer prep) | 52 weeks |
| Water storage & distribution systems | 36–44 weeks |
| Process vessels & mixing systems | 42 weeks |
Lead times vary based on system complexity, scope, and project requirements.
Engineering & Project Delivery Capacity
-
ZETA supports large-scale biopharma projects with global engineering resources and integrated fabrication capabilities.
- 2.5 million project hours delivered annually
- Coordinated engineering, automation, and fabrication across global teams
- Proven ability to execute multiple complex projects in parallel.
Lets talk about your next project
If you’re evaluating a specific project, we can provide a more precise timeline and budget based on your process, scope, and facility requirements.
About ZETA USA
ZETA USA delivers integrated process engineering, automation, and precision fabrication for biotech and biopharma. From simulation and process design to turnkey skids and super-skids, our systems are built to perform without rework or surprises. With more than 35 years of global experience and a proven fabrication methodology, we help manufacturers and A&E partners reduce project risk and accelerate time-to-market.
| © 2025 ZETA USA | 2200 Renaissance Blvd, Ste 170 | King of Prussia, PA 19406 |


